Viral suppression data
See the dataCure in 8 Weeksa,b
The First and Only FDA-Approved DAA For Both Acute and Chronic HCV1
Cure = SVR12 (HCV RNA <LLOQ 12 weeks after the end of treatment).
aFor TN, NC and CC patients.
bLiver or kidney transplant recipients are not eligible for an 8-week regimen.
AI-generated imagery. Not real patients.
MAVYRET is indicated for the treatment of adult and pediatric patients 3 years and older with acute or chronic hepatitis C virus (HCV) genotype 1, 2, 3, 4, 5, or 6 infection without cirrhosis or with compensated cirrhosis (Child-Pugh A). MAVYRET is indicated for the treatment of adult and pediatric patients 3 years and older with HCV genotype 1 infection, who previously have been treated with a regimen containing an HCV NS5A inhibitor or an NS3/4A protease inhibitor (PI), but not both.1
Acute HCV1,2
96%
Cure Rate
(n=275/286)
ITT
100%
Cure Rate
(n=275/275)
mITT
- 0% on-treatment virologic failure (n=0/286)
- 0% relapse (n=0/286)
Single-arm, open-label study of GT 1-4, TN, NC and CC adult participants with acute HCV treated for 8 weeks.
Chronic HCV3
98%
Cure Rate
(n=1218/1248)
ITT
99%
Cure Rate
(n=1218/1226)
mITT
- 0.1% on-treatment virologic failure (n=1/1248)
- 0.6% relapse (n=7/1226)
Integrated, pooled analysis of GT 1-6, TN, NC and CC adult participants with chronic HCV treated for 8 weeks from 8 clinical trials.
See clinical trial study designs below.
MAVYRET is contraindicated in patients with moderate or severe hepatic impairment (Child-Pugh B or C) or those with any history of prior hepatic decompensation.1
Cure HCV at the First Detectable Viral Load
Cure = SVR12 (HCV RNA <LLOQ 12 weeks after the end of treatment).1
Relapse = HCV RNA ≥LLOQ after end-of-treatment response among participants who completed treatment.1
mITT population excludes participants who did not achieve SVR12 for reasons other than virologic failure.1
aIn TN, NC and CC patients.
bLiver or kidney transplant recipients are not eligible for an 8-week regimen.
Viral Suppression Across 8 Weeks14
In Non-Cirrhotic Patients With Chronic HCV Completing 8 Weeks of MAVYRET
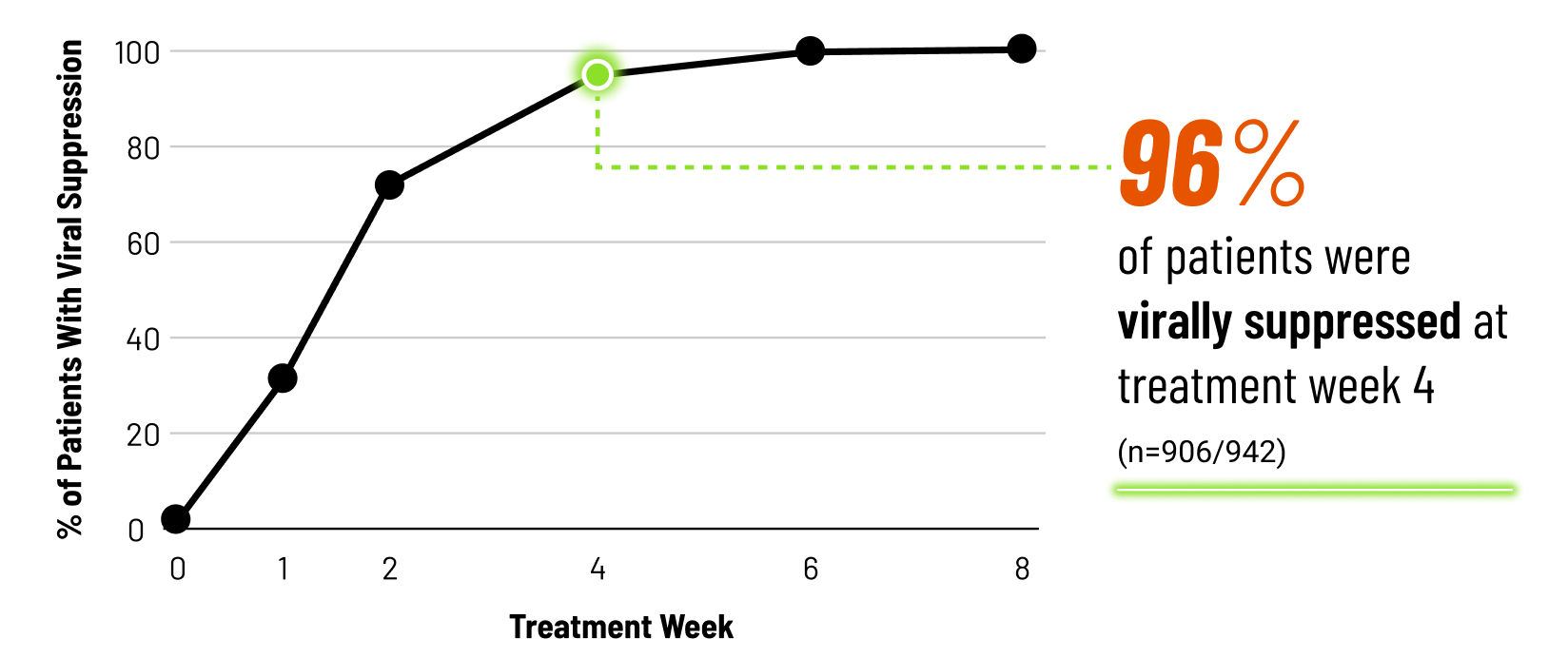
Viral suppression at treatment week 4 is not a clinical endpoint for cure.
METHODOLOGY14
Post hoc exploratory pooled analysis from 5 phase 2/3 clinical trials of chronic HCV GT 1-6, TN or PRS-TE, NC adult patients (N=950) treated with MAVYRET for 8 weeks. HCV RNA was measured at baseline, treatment weeks 1, 2, 4, 5, and at the end of treatment. After excluding patients lost to follow-up or missing SVR12 data (n=13) or with on-treatment virologic failure (n=2), 942 had data on viral suppression (defined as HCV RNA <LLOQ).
LIMITATIONS1,14
Product labeling for treatment duration should be followed regardless of HCV RNA levels at treatment week 4. The included clinical studies were not powered to assess the impact of on-treatment viral suppression on SVR12. No conclusions should be made from this exploratory analysis.
Due to assay difference across studies, LLOQ was 25 IU/mL (Roche COBAS TaqMan® RT-PCR assay v. 2.0) or 15 IU/mL (Roche COBAS Ampliprep/TaqMan® RT-PCR assay v. 2.0).
Cure = SVR12 (HCV RNA <LLOQ 12 weeks after the end of treatment).1
Relapse = HCV RNA ≥LLOQ after end-of-treatment response among participants who completed treatment.1
mITT population excludes participants who did not achieve SVR12 for reasons other than virologic failure.1
aIn TN, NC and CC patients.
bLiver or kidney transplant recipients are not eligible for an 8-week regimen.
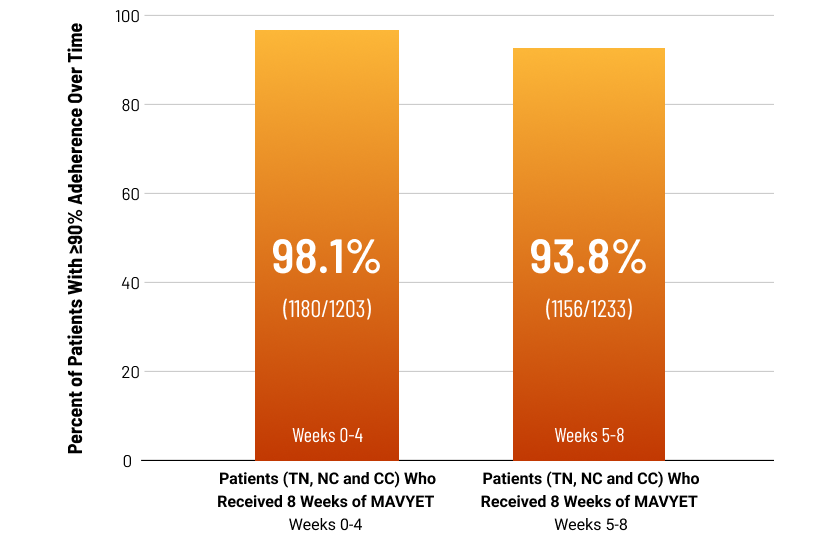
Cure Rates in Patients With Varied Adherence15
Based On a Retrospective, Exploratory Analysis of NC and CC Patients From Ten Phase-3 Clinical Trials
100%
Cure Rate With
<90% Adherence (mITT SVR12)
During weeks 0-4 (n=21/21) and weeks 5-8 (n=76/76)
99%
Cure Rate With
≥90% Adherence (mITT SVR12)
During weeks 0-4 (n=1155/1162) and weeks 5-8 (n=1136/1143)
Percent of Patients With ≥90% Adherence Over Time
Tell your patients that it is important not to miss or skip doses of MAVYRET during treatment.
METHODOLOGY15
Post hoc, exploratory analysis pooled data from 10 phase 3 clinical trials evaluating 8-week treatment with MAVYRET in chronic HCV GT 1-6, TN, NC and CC adult patients. Endpoints included SVR12 and adherence, calculated as the percentage of pills taken relative to the total number expected to be taken during each dispensation interval [weeks 0-4 and weeks 5-8], excluding patients with missing pill count data for that interval.
LIMITATIONS15
The included clinical studies were not powered to assess differences between groups or the impact of adherence on SVR12; therefore, conclusions from this exploratory analysis should be drawn with caution. Patients with stage 4 or 5 chronic kidney disease, or with prior HCV treatment, were excluded. Patients with missing pill count data were excluded for each interval.
SELECT BASELINE CHARACTERISTICS15
- Stable opioid substitution therapy: 9% (n=111/1304)
- History of psychiatric disorder: 27% (n=349/1304)
- ≥5 concomitant medications: 27% (n=351/1304)
- Currently using alcohol: 33% (n=435/1304)
- History of injection drug use: 39% (n=510/1304)
Patients with a history of psychiatric disorders were self-reported or defined as those with reported concomitant psychiatric medications. Patients using alcohol or with any history of injection drug use were self-reported.
Patient subgroups are not mutually exclusive.

The ONLY 8-WEEK Pangenotypic Cure For Your TN, NC and CC Patients1,a,b
See All MAVYRET Patient PopulationsCure = SVR12 (HCV RNA <LLOQ 12 weeks after the end of treatment).1
Relapse = HCV RNA ≥LLOQ after end-of-treatment response among participants who completed treatment.1
mITT population excludes participants who did not achieve SVR12 for reasons other than virologic failure.1
aIn TN, NC and CC patients.
bLiver or kidney transplant recipients are not eligible for an 8-week regimen.



